
Orvida pharma
Disease-Modifying Therapies for Rare Genetic Skin Diseases

The Unmet Needs in Orphan Rare Skin Diseases
Palmoplantar Epidermal Differentiation Disorders (pEDD), Keratoderma, Ichthyosis
.png)
Chronic, early-onset diseases
Triggered by skin dysfunction, beginning in infancy and increasing in severity throughout life.
Triggered by skin dysfunction, beginning in infancy and increasing in severity throughout life.
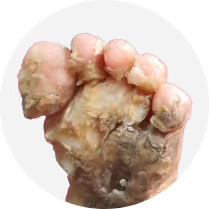
Severe, devastating manifestations Causing significant pain, open lesions, mutilations, impaired mobility and other debilitating complications.
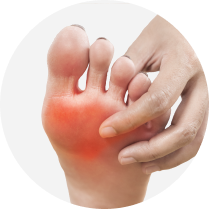
No disease-modifying
therapies available
Current treatments only attempt to manage the symptoms.
Current treatments only attempt to manage the symptoms.

A Clinical-Stage Biotech Focused on Rare Skin Diseases
Orvida Pharma is a clinical-stage company developing disease-modifying therapies for rare, severe skin diseases.
Our oral and topical TRPV3 inhibitor platform target conditions including Olmsted Syndrome, Palmoplantar Keratoderma (pEDDs), and Ichthyosis, aiming to deliver durable solutions for patients with significant unmet needs.
Our Pipeline
Discovery
Preclinical
Phase Ia
Phase Ib
in patients
Phase 2
Phase 3
Marketing approval
Olmsted Syndrome (TRPV3-pEDD)
Ontivatrep (oral)
Ontivatrep (oral)
Pachyonychia Congenita (pEDD-PC)
Ontivatrep (oral)
Ontivatrep (oral)
Ichthyosis
Ontivatrep (oral)
Ontivatrep (oral)
Keratoderma (pEDD)
KM-001 (topical)
KM-001 (topical)
pEDD = palmoplantar Epidermal Differentiation Disorder
Clinical Trial: Open and Recruiting
Clinical Trial:
Open and Recruiting
Our clinical trial with an oral drug for Olmsted Syndrome (TRPV3-pEDD) is currently ongoing.
Additional studies in other rare skin conditions are expected to open later this year - stay tuned.
Contact Us
Thank You
We will contact you as soon as possible
Oops! Something went wrong while submitting the form.

