Science
Targeting a Shared Molecular Driver in Rare Skin Diseases: TRPV3
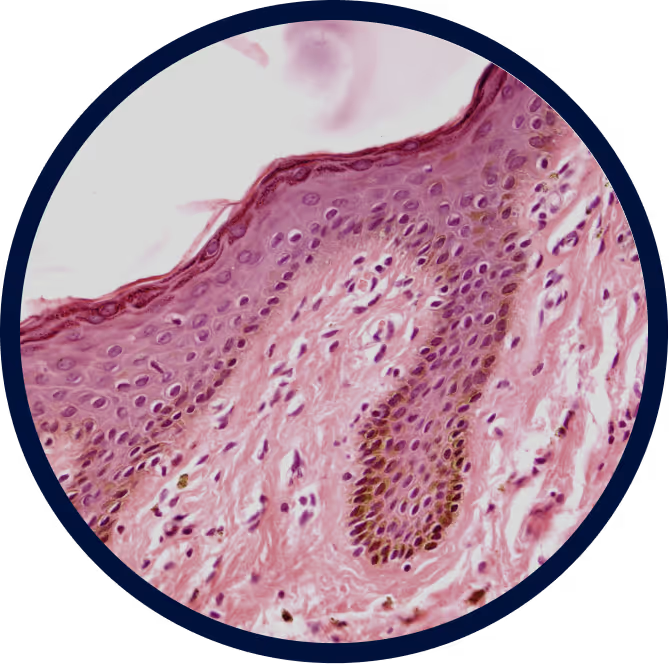
Transient Receptor Potential Vanilloid-3 (TRPV3):
one target, multiple rare skin diseases
Mutations and over expression of TRPV3, a calcium-permeable ion channel expressed in keratinocytes, lead to channel hyperactivation and dysregulated calcium influx in the epidermis. This calcium imbalance disrupts keratinocyte differentiation and skin barrier integrity, triggering inflammation, hyperkeratosis, and progressive skin dysfunction characteristic of pEDD disorders such as Olmsted Syndrome, Pachyonychia Congenita, severe Keratodermas and Ichthyosis. The resulting epidermal instability drives early-onset, chronic disease marked by painful lesions, impaired mobility, and cumulative tissue damage.
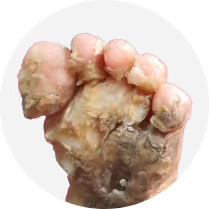
Olmsted Syndrome
.avif)
Pachyonichia Congenita
.png)
Ichthyosis

Inhibiting TRPV3,
Normalizing Calcium Levels, Restoring Healthy Skin
Inhibiting TRPV3 normalizes calcium influx in keratinocytes, restoring proper skin cell function and barrier integrity. This targeted approach addresses the root cause of these skin disorders, reducing inflammation, hyperkeratosis, pain and the progression of skin disease.
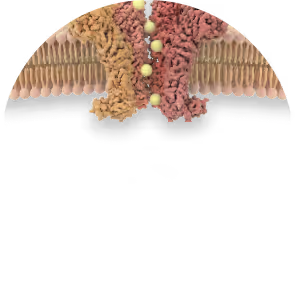
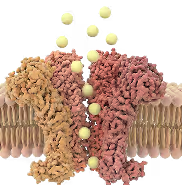
TRPV3
(calcium channel)
hyper activation
hyper activation
Calcium influx dysregulation
Skin
dysfunction
dysfunction
Disease onset
Lesions, pain,
disabilities
disabilities

TRPV3
inhibition
inhibition
Ontivatrep
(Oral)
(Oral)
KM-001
(Topical)
(Topical)
Calcium
homeostasis
homeostasis
Healthy
skin
skin
Disease
mitigation
mitigation
Bringing Disease-Modifying Therapies to Life
Orvida’s patient-centered clinical strategy focuses on palmoplantar epidermal differentiation disorders (pEDDs) such as Olmsted Syndrome, Keratoderma, and Ichthyosis, as well as related rare skin diseases. Clinical efficacy and safety have been demonstrated in clinical studies of a topical therapy, while Phase I safety study of the oral TRPV3 inhibitor ontivatrep has been completed. A Phase Ib study in Olmsted Syndrome is ongoing, with a pivotal study planned to follow. Expansion into additional TRPV3 related skin indications will take place in the near future.