
Pipeline
Focused Science. Broad Patient Impact. Global Outreach.
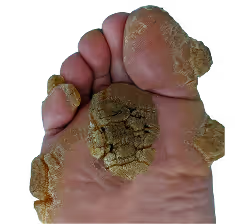
Olmsted Syndrome is an ultra-rare genetic skin disease marked by severe thickening of the palms and soles, painful lesions, and inflammation causing sever mobility challenges, starting in early childhood. OS is caused by gain-of-function mutations in the TRPV3 ion channel.
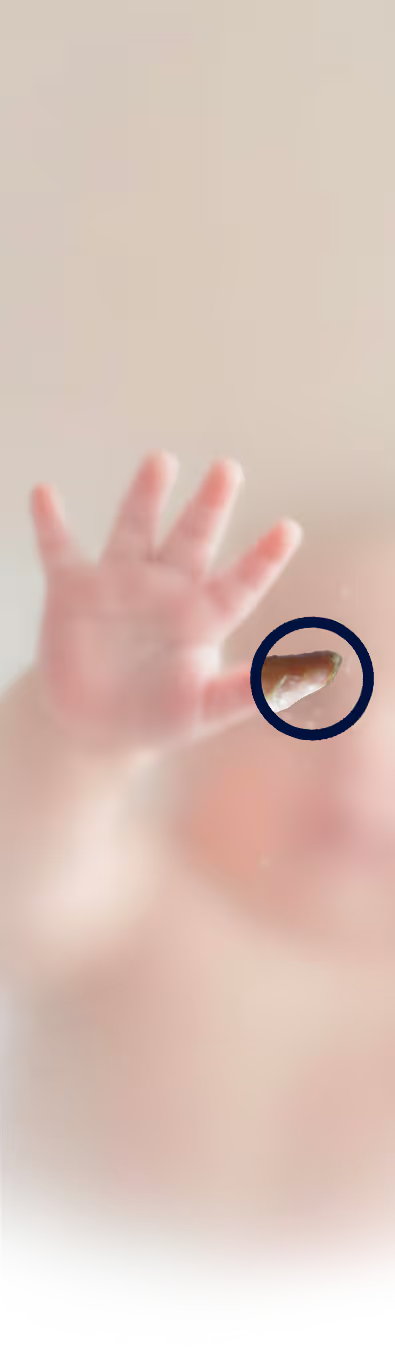
View DataPachyonychia Congenita is a rare inherited skin disorder characterized by painful plantar calluses and characteristic nail changes that can limit walking and daily activities. PC is caused by mutations in keratin genes, and over expression of TRPV3 resulting in increased TRPV3 signaling.
View DataIchthyosis is a group of rare, severe inherited skin disorders characterized by dry, thickened, scaly skin caused by an impaired skin barrier. It is driven by mutations in genes involved in barrier formation and lipid processing, and can range from mild scaling to thick, rigid plates covering the entire body with widespread cracking affecting the face (inverted eyelids/lips) and restricting limb movement.
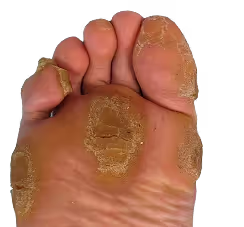
View DataKeratoderma (most commonly Palmoplantar Keratoderma, PPK) is a group of genetic skin diseases characterized by abnormal thickening of the skin on the palms and soles. It is caused by mutations in genes involved in keratin structure and epidermal differentiation, leading to painful fissures, inflammation, and impaired mobility that can significantly affect daily function.
View DataLanavatrep (Oral KM-023)




Lanavatrep (Oral)
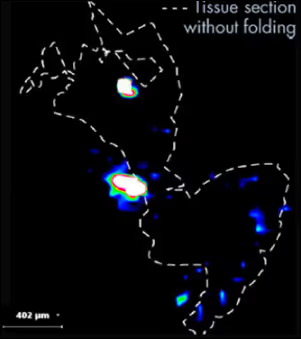
- Lanavatrep reaches the skin within just four hours, achieving rapid target engagement.
- Sustained exposure over 24 hours in healthy volunteers supports the potential for durable efficacy.
Lanavatrep (Oral)
.avif)
3D culture
.avif)
3D skin culture
.avif)
3D skin 250nM KM-023
KM-001 (Topical)